CRISPR gene editing shows promise for sickle cell disease
Trials have been effective but the treatment is extremely expensive, for now.
A breakthrough in CRISPR gene editing for patients with blood diseases shows the promise and problems with the new technology, according to the New England Journal of Medicine (as seen by Science Mag). Researchers from CRISPR Therapeutics engineered DNA-cutting enzyme that forced blood cells to produce more hemoglobin, allowing patients to live pain free without the need for regular blood transfusions. However, treatments could cost in the millions of dollars, meaning it won't help many folks until the prices fall significantly.
Researchers focused on two conditions, beta-thalassemia and sickle cell anemia, the latter which affects upwards of 100,000 Black people in the United States alone. Sickle cell disease causes flexible red blood cells to stiffen and form a crescent-moon shape, causing patients severe pain and boosting the risk of organ damage, stroke and other problems.
It's wonderful, but it won't solve the global health problem.
In severe cases, patients receive bone-marrow transplants, a risky treatment that requires perfectly matched donors that are very difficult to find. While the CRISPR treatment also requires a bone marrow transplant, it uses the patient's own blood stem cells.
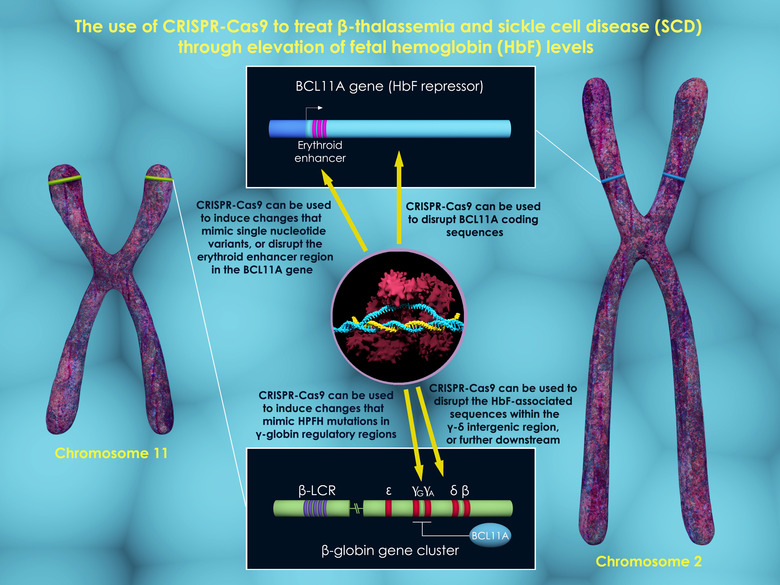
First, blood stem cells were extracted from the patient and the defective cells destroyed using chemotherapy, much like with regular bone marrow transplants. Then, researchers used CRISPR's DNA-cutting enzyme's and guide RNA to break BCL11A — a genetic switch that shuts off the gene for fetal hemoglobin early in life. With that gene now reactivated, fetal hemoglobin is produced that restores missing missing hemoblogin for beta-thalassemia patients and replaces flawed hemoglobin in sickle cell patients.
As a result, most patients in the trials made sufficient amounts of fetal hemoglobin and no longer needed blood transfusions. Those include a teenager who can now swim pain-free, along with a young man who has gone without blood transfusions for two and half years after needing them regularly before, according to Boston Children's Hospital.
Researchers need to follow up for longer periods to make sure there are no side effects like cancer that can be caused by CRISPR. However, the biggest issue is the cost — though the team didn't mention the potential price of treatment, a comparable non-CRISPR gene-therapy option may cost as much as $1.8 million per patient, including the chemotherapy and hospital stay. That obviously rules out a lot of patients, particularly in the US where health care costs can be astronomical.
However, the National Institute of Health and the Bill and Melinda Gates Foundation are injecting up to several hundred million dollars into the tech in hopes of reducing the costs. That could include new treatment methods that would eliminate the need for a bone marrow transplant. "It's a big hairy goal... but we'll get there," said gene University of California, Los Angeles researcher Donald Kohn.