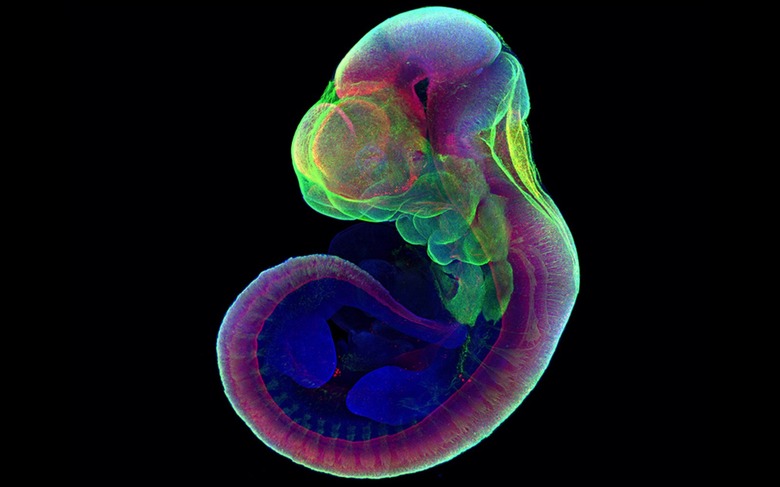
This mouse embryo grew in an artificial uterus
It was previously believed a fetus couldn't survive without a living womb.
We may receive a commission on purchases made from links.
Scientists have managed to grow the embryo of a mammal outside of the womb for the first time. In a study published on Wednesday in Nature, a team of researchers from the Weizmann Institute of Science in Israel say they've successfully grown more than 1,000 mice embryos for six days using a process that involves a mechanical device. The first part of the experiment saw the team remove the mice from their mothers' wombs after five days. In an interview with The New York Times, Dr. Jacob Hanna, one of the researchers on the project, said his team has since managed to take an embryo from a female mouse right after fertilization and grow it for 11 days. What's more, the lab-grown embryos are consistently identical to their 'real' counterparts.
The team spent seven years making the machine that has enabled their research. It's a two-part system that consists of an incubator and ventilation system. Each of the embryos floats in a vial that's filled with a special nutrient-laden fluid. A wheel gently spins the mice so that they don't attach to the wall of their temporary home. This prevents the embryos from deforming and subsequently dying. Meanwhile, the attached ventilator provides the mice with oxygen while maintaining their environment's flow and pressure.
It takes about 20 days for a mouse to gestate to the point where it can survive outside the womb. So far, the mechanical womb Dr. Hanna and his team have created can sustain the mice through 11 days of growth. It's at that point, in what would be more than halfway through a regular pregnancy, that the mice die off. The embryos become too large to survive on merely the nutrients they absorb through diffusion. They need a blood supply, and that's the next technical challenge the team plans to solve. One potential solution on the table includes an artificial blood supply that could connect to mice's placentas, Dr. Hanna told The NY Times.
Before you run for the hills, know that Dr. Hanna's team didn't create the device to disrupt nature's natural order. Instead, they're using their process to study how factors like genetic mutations and environmental conditions can affect the growth of a fetus while inside the womb. Until this breakthrough, scientists had turned to species like worms and frogs — that is, non-mammals — to study the development of tissues and organs. A similar device may one day allow scientists to grow a human baby in the same way, but that's something that's years and decades away, provided it's even possible.