treatment
Latest

Microsoft blood bot helps recovered COVID-19 patients donate plasma
Microsoft’s Plasma Bot is a screening tool meant to help recovered COVID-19 patients donate their plasma to treatment efforts.

IBM shares AI tools to better understand and treat COVID-19
It has released a series of new tools to aggregate data, help researchers explore potential therapies, advance the study of newly sequenced SARS-CoV-2 genomes and make the latest info available to healthcare professionals. Its AI deep search tool is ingesting data from the White House, a coalition of research groups and licensed databases from the DrugBank, Clinicaltrials.gov and GenBank. The cloud-based research tool is a repository of genes, proteins and other molecules from sequenced viral and bacterial organisms, and it’s meant to help researchers identify molecular targets necessary for drug design, test development and treatment.
AI-formulated medicine to be tested on humans for the first time
A drug designed entirely by artificial intelligence is about to enter clinical human trials for the first time. The drug, which is intended to treat obsessive-compulsive disorder, was discovered using AI systems from Oxford-based biotech company Exscientia. While it would usually take around four and a half years to get a drug to this stage of development, Exscientia says that by using the AI tools it's taken less than 12 months.
Microsoft backs AI in healthcare with a $40 million program
Microsoft's deepening health tech strategy now includes a significant commitment to AI. The company is starting a $40 million AI for Health program that should help researchers and key organizations improve the quality of life for people around the planet. The five-year initiative will use AI to improve diagnosis, prevention and treatment, as you might have expected, but it'll also be used for global-scale health insights and to improve access to healthcare in areas where it's often costly or hard to find.

Google bans ads for unproven medical treatments
In an attempt to remain trustworthy, Google will no longer allow ads for "unproven or experimental medical techniques." Under the new Healthcare and medicines policy, the company will prohibit advertising for treatments that lack biomedical or scientific basis, including most stem cell therapies and gene therapy. That should cut down on ads for things like young blood transfusions and vampire facials.

GSK to use 23andMe’s DNA library in drug development
DNA testing company 23andMe has partnered with pharmaceutical giant GlaxoSmithKline (GSK), in a bid to develop new drug treatments. 23andMe, which gives customers insight into their genetic makeup via postal saliva tests, has some five million customers -- a potential DNA database considerably larger than those generally available to the scientific community. "By working with GSK, we believe we will accelerate the development of breakthroughs," 23andMe CEO Anne Wojcicki wrote in a blog post.

Google will pre-vet addiction treatment ads following scams
In September, following a Verge report detailing scams conducted through misleading drug and alcohol treatment advertising on Google, the company began pulling addiction treatment-related ads in the US. In January, Google did the same in the UK following a similar report from The Sunday Times and later, it extended the suspension of these sorts of ads globally as it worked out a strategy to ensure those advertising on its platform were legitimate. Now, Reuters reports, Google has established a way to vet addiction treatment facilities wanting to post ads.

NASA tests treatment that could slow astronauts’ muscle loss
Even though astronauts exercise everyday while they're aboard the International Space Station (ISS), they still experience a fairly significant loss of muscle mass. So much so that they require physiotherapy once they return to Earth. However, researchers with the Houston Methodist Research Institute and the Novartis Institute for Biomedical Research are testing a device that can continuously administer a medicine that may help prevent muscle deterioration, Seeker reports. The team sent 40 mice to the ISS in December, some with devices that deliver the drug and some with devices that just deliver an inactive solution. Half returned to Earth in January and the other half will do so this month.

Researchers treat ear defect by growing implants from cells
While scientists have been working on growing organs and body parts like ears for some time, researchers in China have taken it one important step further. They've grown new ears for five children with microtia -- an ear defect that results in small, underdeveloped ears and often affects hearing -- grafted them onto the children and continued to observe the development for over two years. The work is important because it's the first study of its kind and shows that these sorts of methods stand to be a viable option going forward.

AI-powered robot finds common soap ingredient may combat malaria
Around half of the world's population is at risk of contracting malaria and it causes around half a million deaths each year. However, the parasites that cause malaria are becoming more resistant to the drugs we currently use to combat them, meaning the global malaria risk stands to increase if we don't develop new drugs quickly enough. Well new research published recently in Scientific Reports finds that a common chemical used in everything from soap and toothpaste to clothing and furniture might be an effective treatment, and it was done with the help of AI.

3D-printed bacteria ink could be used to treat burns
In a new study published today in Science Advances, researchers present a 3D-printable ink that contains bacteria and they say that depending on what species of bacteria it holds, the ink stands to have a number of useful applications. "Printing using bacteria-containing hydrogels has enormous potential, as there is such a wide range of useful bacteria out there," Patrick Rühs, an author of the study, said in a statement.
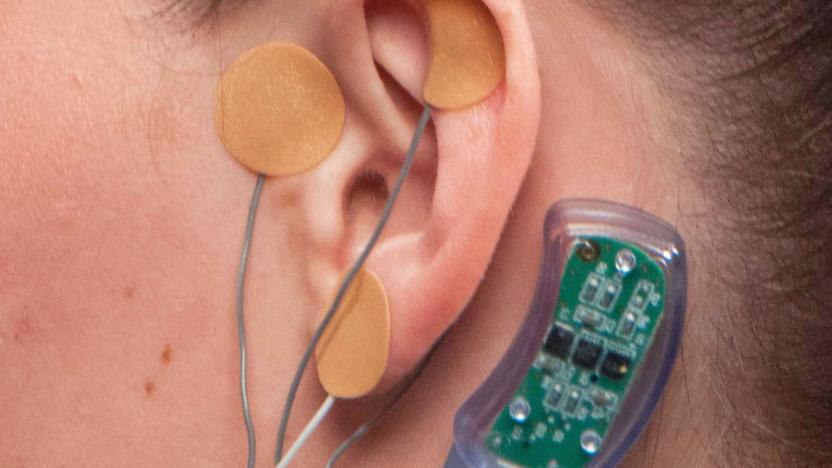
FDA approves an electrical device to help ease opioid withdrawal
Amid an epidemic of opioid addiction, doctors are looking for ways to help people wean themselves off of the drugs, but withdrawal symptoms are a major hurdle. Innovative Health Solutions says its NSS-2 Bridge is a "percutaneous nerve field stimulator (PNFS) device system" that sends electrical pulses to certain cranial nerves, treating symptoms including sweating, tremors, stomach upset, joint pain and anxiety. Yesterday the FDA cleared it for marketing, making this the first device approved for use in this way. Like the DEKA Arm System, this device was reviewed through the agency's de novo pathway that fast-tracks "some low- to moderate-risk devices." Prior to approval, the FDA reviewed a clinical study of 73 patients where all of them showed at least a 31 percent drop in their clinical opiate withdrawal scale (COWS) score within 30 minutes of use.

Navajo Nation may undo genetic research ban in hopes of better care
The Navajo Nation banned genetic studies in 2002 due to concerns over how its members' genetic material would be used, but, as Nature News reports, the Navajo are considering a reversal of that policy. An oncology center is set to open next year on Navajo lands and the tribe's research-ethics board is looking into allowing some genetic research to take place at the facility.

A nanoparticle-coated skin patch could treat obesity and diabetes
A new study out today in ACS Nano presents an interesting and effective way to reduce fat stores in the body. Researchers at Columbia University and the University of North Carolina showed that a patch loaded with nanoparticles could reduce fat, increase energy expenditure and ameliorate type-2 diabetes in obese mice.
Microscope advances could prevent multiple breast cancer surgeries
A new type of microscope could drastically reduce the number of women having multiple breast cancer surgeries, researchers at the University of Washington claim. Until now, there's been no reliable way to determine whether surgeons have completely removed all cancerous tissue during surgery, meaning between 20 and 40 percent of women have to undergo second, third or even fourth procedures.
Prostate cancer laser treatment cures half of trial subjects
A new prostate cancer treatment that combines lasers and deep-sea bacteria could be "truly transformative," according to a team of researchers. A trial conducted with 415 men across Europe finished with nearly half completely free of cancer compared to 13.5 percent in a control group To top it off, unlike with current, aggressive therapies that can cause impotence and urinary problems, most of the subjects were free of side effects after two years.

FDA approves new trials using MDMA to help treat PTSD
MDMA, the pure form of Ecstasy, is usually only mentioned when partygoers get busted for holding the Schedule 1 restricted drug. But medical researchers have been quietly testing its potential medical applications, like treating terminally ill patients and assisting patient therapy for PTSD. The results from the latter have been promising enough for the FDA to commission Phase 3 trials, which is the last step before MDMA's possible approval as a prescription drug.

Graphene key to promising treatment for spinal cord injuries
Graphene seems to have almost limitless potential, from making better batteries to night-vision windshields and microscopic sensors. And now, a team at Rice University has shown the material could be key to a promising new treatment for severe spinal cord injuries.

Scientists use light to nuke cancer cells in mice
In the great fight against cancer, scientists look for solutions that are both effective and less ravaging than current treatments like chemotherapy. One experimental concept, optogenetics, uses light to reduce or eliminate cancer cells. Researchers at UT San Antonio have devised a method using this school of treatment to attack inoperable or hard-to-reach tumors, which could give options to patients who were considered too high of risk to help.
Experimental Zika vaccine approved for human trials
The Food and Drug Administration approved an experimental Zika vaccine called GLS-5700 for a clinical trial in humans earlier today, the first such treatment to get an official nod from the agency. Initial trials will start with 40 healthy subjects getting dosed in the coming weeks, and if all goes without a hitch, preliminary results should be available later this year.








