Researchers grew a fully mature human egg in a lab
It could help fertility treatments in the future.
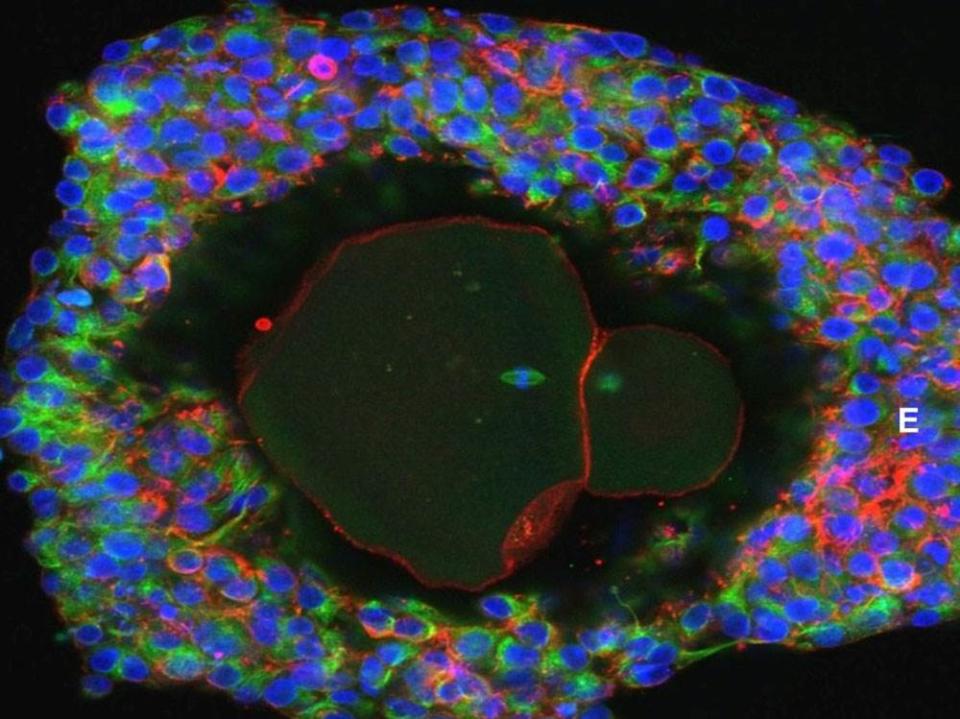
Researchers in the UK and the US have taken human eggs in their most early stage and developed them to maturity in a lab for the first time. It's a big achievement that could open up new avenues for infertility treatment and give scientists a better understanding of how the egg development process works and how it can go wrong. While this has been done before in mice, experiments of which have resulted in live offspring, this is the first time it has been done with human eggs.
The research team had previously developed a protocol wherein they could take eggs in later stages of development and bring them to maturity, and they expanded that protocol in an attempt to develop eggs from start to finish in the lab. They took samples of ovarian tissue from 10 women while they were undergoing caesarean section surgery and cultured sections of that tissue that have the ability to release an egg, structures known as follicles. The follicles were cultured in a multi-step procedure that allowed for eggs to develop. At the end of each step, follicles, and eventually eggs, that had successfully matured further were moved to the following step. The team initially isolated 87 follicles and by the end of the 21-day procedure, nine eggs reached a stage of maturity that would in theory allow them to be fertilized with sperm and develop into an embryo.
While only 37 percent of the early-stage follicles resulted in a fully grown egg and only 10 percent achieved levels of maturity that could allow for reproduction, it's still an important step in the study of egg development, or oogenesis. Ali Abbara, an endocrinology senior lecturer at the Imperial College of London who wasn't involved with the study, told Reuters, "The technology remains at an early stage, and much more work is needed to make sure that the technique is safe and optimized before we ascertain whether these eggs remain normal during the process, and can be fertilized to form embryos that could lead to healthy babies." Evelyn Telfer, a researcher at the University of Edinburgh and the leader of the study, told Reuters that the team is now working on optimizing this process and evaluating how healthy the resulting eggs are. One next step is to try to fertilize the eggs with sperm to see if they can result in a viable embryo -- work that requires a licence that the team doesn't yet have.
This work could help improve IVF treatments in the future and could particularly help girls with cancer. They typically have pieces of their ovaries removed and preserved prior to receiving cancer treatments that might damage their follicles. However, if there's a chance that the tissue has cancer cells, it can't be reimplanted later on. If doctors could mature those eggs outside of her body, those eggs could still be used without introducing risk to the woman. Additionally, in cases where women have a condition that prevents the development of their eggs, this type of research could help scientists understand why that happens. "There are several conditions in which women lose their eggs much earlier in life, or they don't grow," Telfer told New Scientist. "If we could understand the process...we could develop treatments for that."
The research was published in Molecular Human Reproduction.