US medical panel recommends 3-person embyronic testing
Calm down, we aren't making chimera-babies. Yet.
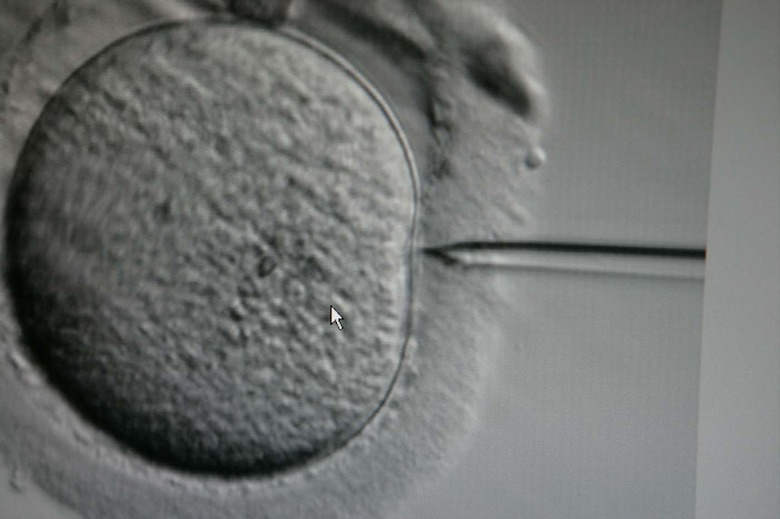
The US National Academy of Medicine recommended Wednesday that the Federal Drug Administration should approve clinical trials for so-called "3-parent embryos." It's a controversial procedure that would transfer mitochondria from a healthy, secondary human egg donor into a fertilized diseased embryo.
Mitochondria are organelles that generate a cell's operational energy. However, unlike the multi-million base-paired DNA within in the cell's nucleus, mitochondrial DNA only contains about 16,000 base pairs. That makes it far more susceptible to dangerous mutations. These mutations can increase a person's chances of suffering everything from migraines to cardiac and respiratory failure. By swapping out the mitochondria, researchers hope to repair the damaged exo-nucleic DNA and prevent the diseases they cause from ever occurring. However, given how new and relatively untested the technique is, there are a ton of safety, technological concerns — not to mention the social, psychological and political implications for people with three parents.
Still, the medical benefits outweigh the risks enough to at least take a closer look at the technology, according to the NAM. The medical panel suggests that the technique only be applied to male embryos (since mitochondrial DNA is only passed to female offspring). It also recommends monitoring the resulting people for years afterward and sharing any data garnered with them. Should the male-only initiative be proven safe, the NAM panel recommends only then expanding it out to female embryos. This policy would stand in stark contrast to guidelines passed in the UK last year which OK'd mitochondrial replacement techniques for both sexes of embryos.